|
Create a Team with knowledge of regulatory guidelines/compliance, validation procedures, laboratory processes, and the technology.Develop Clear and Precise Functional and User Requirements.In the United States, for example, the FDA requires pharmaceutical companies to perform CSV for systems that support the production of:īest Practices for Computer System Validation  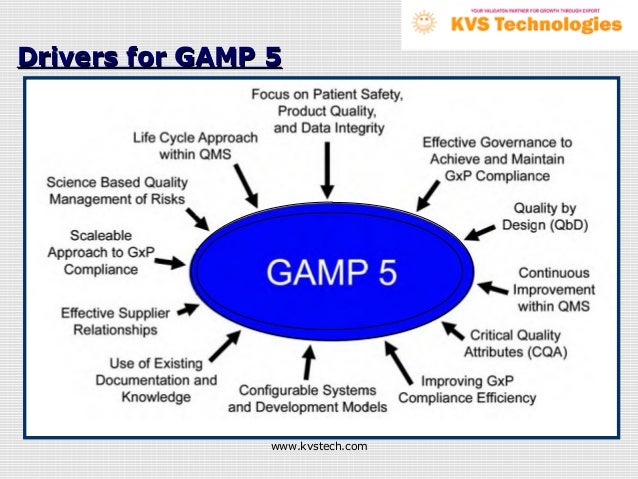
These regulatory agencies require CSV processes to confirm the accuracy and integrity of data in computerized systems in order to ensure product safety and effectiveness. Good Automated Manufacturing Practice (GAMP 5 ®) sets out principles and procedures that help ensure that pharmaceutical or medical device products have the required quality and details a recognized standard for Computer system validation (CSV).Ĭomputer System Validation (CSV) is a documented process that is required by regulatory agencies around the world to verify that a computerized system does exactly what it is designed to do in a consistent and reproducible manner. GAMP® 5 provides pragmatic and practical industry guidance to achieve compliant computerized systems fit for intended use in an efficient and effective manner.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
